Ruth Eckstein Grunau
PhD
Investigator Emerita, BC Children's Hospital


NMR in Biomedicine
Zhu, C. and Chan, N. and Chacko, A. and Holsti, L. and Grunau, R. and Weber, A.
DOI: 10.1002/nbm.70065Pediatric Research
Carmichael, T.G. and Rauscher, A. and Grunau, R.E. and Weber, A.M.
DOI: 10.1038/s41390-025-03966-6Biological Psychiatry
McLean, M.A. and Weinberg, J. and Vinall Miller, J. and Guo, T. and Synnes, A.R. and Miller, S.P. and Grunau, R.E.
DOI: 10.1016/j.biopsych.2025.06.005Journal of Pediatrics
Ybarra, M. and Chau, V. and Au-Young, S.H. and Raghuram, K. and Guo, T. and Grunau, R.E. and Branson, H. and Shah, V. and de Vries, L.S. and Miller, S.P. and Cizmeci, M.N.
DOI: 10.1016/j.jpeds.2025.114713Journal of Pediatrics
Ybarra, M. and Selvanathan, T. and Guo, T. and Chau, V. and Branson, H.M. and Ly, L.G. and Synnes, A.R. and Kelly, E. and Grunau, R.E. and Miller, S.P. and Tam, E.W.Y.
DOI: 10.1016/j.jpeds.2025.114503Journal of Pediatrics
Oloomi, S. and Ufkes, S. and Selvanathan, T. and Chau, C. and Roland, E. and Guo, T. and Chau, V. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2025.114667Obstetrics and Gynecology
Liauw, J. and Campbell, K.S.J. and Foggin, H. and Grunau, R.E. and Petrie, J. and Qasim, A. and Brignardello-Petersen, R. and Mishaal, R.A. and Hutcheon, J.A.
DOI: 10.1097/AOG.0000000000005950Developmental Psychobiology
McLean, M.A. and Weinberg, J. and Synnes, A.R. and Miller, S.P. and Grunau, R.E.
DOI: 10.1002/dev.70064Journal of Pediatrics
Christensen, R. and Chau, V. and Synnes, A. and Guo, T. and Ufkes, S. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2024.114271JAMA Network Open
McLean, M.A. and Ranger, M. and Bone, J.N. and Selvanathan, T. and Au-Young, S.H. and Chau, C.M.Y. and Chau, V. and Ly, L. and Kelly, E. and Synnes, A. and Miller, S.P. and Grunau, R.E.
DOI: 10.1001/jamanetworkopen.2025.4477Journal of Pediatrics
Kim, C. and Ufkes, S. and Guo, T. and Chau, V. and Synnes, A. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2025.114524Pediatric Research
El Shahed, A. and Chau, V. and Terry, J. and Whitehead, C. and Synnes, A. and Grunau, R. and Miller, S.P.
DOI: 10.1038/s41390-025-03945-xNeurology
Selvanathan, T. and Oloomi, S. and Guo, T. and Ufkes, S. and Chau, V. and Branson, H. and Synnes, A. and Ly, L.G. and Kelly, E.N. and De Vries, L.S. and Grunau, R.E. and Miller, S.P.
DOI: 10.1212/WNL.0000000000214016Journal of Pediatrics
Selvanathan, T. and Guo, T. and Ufkes, S. and Chau, V. and Branson, H.M. and Synnes, A.R. and Ly, L.G. and Kelly, E. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2024.114090JAMA Network Open
Selvanathan, T. and Ufkes, S. and Guo, T. and Chau, V. and Branson, H.M. and Ibrahim, G.M. and Ly, L.G. and Kelly, E.N. and Grunau, R.E. and Miller, S.P.
DOI: 10.1001/jamanetworkopen.2024.2551Cardiology in the Young
Roberts, S.D. and Sananes, R. and Wojtowicz, M. and Seed, M. and Miller, S.P. and Chau, V. and Au-Young, S.H. and Guo, T. and Ly, L. and Kazazian, V. and Grunau, R.E. and Williams, T.S.
DOI: 10.1017/S1047951123004316Neurology
Selvanathan, T. and Guo, T. and Ufkes, S. and Chau, V. and Branson, H. and Synnes, A. and Ly, L.G. and Kelly, E.N. and Grunau, R.E. and Miller, S.P.
DOI: 10.1212/WNL.0000000000209264Annals of Clinical and Translational Neurology
Konrad, J. and Guo, T. and Ufkes, S. and Selvanathan, T. and Sheng, M. and Al-Ajmi, E. and Branson, H.M. and Chau, V. and Ly, L.G. and Kelly, E.N. and Grunau, R.E. and Miller, S.P.
DOI: 10.1002/acn3.52168Child Neuropsychology
McLean, M.A. and Weinberg, J. and Synnes, A.R. and Miller, S.P. and Grunau, R.E.
DOI: 10.1080/09297049.2024.2314958Paediatric and Neonatal Pain
Mia A. McLean and Lisa Nakajima and Cecil M. Y. Chau and Joanne Weinberg and Anne R. Synnes and Steven P. Miller and Ruth E. Grunau
DOI: 10.1002/pne2.1211209 / 2023
Neurology
Duerden, E.G. and Guo, T. and Chau, C. and Chau, V. and Synnes, A. and Grunau, R.E. and Miller, S.P.
DOI: 10.1212/WNL.0000000000207817Pediatric Research
Selvanathan, T. and Zaki, P. and McLean, M.A. and Au-Young, S.H. and Chau, C.M.Y. and Chau, V. and Synnes, A.R. and Ly, L.G. and Kelly, E. and Grunau, R.E. and Miller, S.P.
DOI: 10.1038/s41390-023-02536-yBJPsych Open
Hutchison, S.M. and Brain, U. and Grunau, R.E. and Kuzeljevic, B. and Irvine, M. and M{\^a}sse, L.C. and Oberlander, T.F.
DOI: 10.1192/bjo.2022.623Developmental Psychobiology
Zusman, E.Z. and Chau, C.M.Y. and Bone, J.N. and Hookenson, K. and Brain, U. and Glier, M.B. and Grunau, R.E. and Weinberg, J. and Devlin, A.M. and Oberlander, T.F.
DOI: 10.1002/dev.22425Neurology
Selvanathan, T. and Au-Young, S.H. and Guo, T. and Chau, V. and Branson, H.M. and Synnes, A. and Ly, L. and Kelly, E.N. and Grunau, R.E. and Miller, S.P.
DOI: 10.1212/WNL.0000000000207848Journal of Pediatrics
Christensen, R. and Chau, V. and Synnes, A. and Guo, T. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2023.113401Archives of Disease in Childhood - Fetal and Neonatal Edition
Thiviya Selvanathan and Ting Guo and Eddie Kwan and Vann Chau and Rollin Brant and Anne R Synnes and Ruth E Grunau and Steven P Miller
DOI: 10.1136/archdischild-2020-32139703 / 2022
JAMA Network Open
McLean, M.A. and Scoten, O.C. and Chau, C.M.Y. and Synnes, A. and Miller, S.P. and Grunau, R.E.
DOI: 10.1001/jamanetworkopen.2022.38088Clinics in Perinatology
McPherson, C. and Grunau, R.E.
DOI: 10.1016/j.clp.2021.11.014Journal of neonatal-perinatal medicine
Abou Mehrem, A. and Al Awad, E. and Anninck, K. and Au-Young, S. and Aydinol, N. and Bartmann, P. and Benders, M. and Benlamri, A. and Bolderheij, L. and Celik, Y. and Chan, N. and Chau, C. and Chau, V. and Chen, X. and Chetcuti Ganado, C. and Coetser, A. and Cools, F. and Da Rocha, G. and Deigner, H.-P. and Dereymaeker, A. and Deshmukh, L. and Domonoske, R. and Dossani, S. and Dsouza, J.M. and El Gamal, M. and Eshemokhai, P. and Esser, M. and Fiedrich, E. and Franz, A. and Ghosh, A. and Groenendaal, F. and Grunau, R.E. and Venkata, S.K.R.G. and Hamitoglu, S. and Hellstr{\"o}m-Westas, L. and Irvine, L. and Jansen, K. and Javadyan, A. and Jenkin, G. and Kamanga, N. and Kaur, N. and Keles, E. and Keller, M. and Kelly, E. and Kesting, S.J. and Kgwadi, D. and Kim, B. and Kohl, M. and Kowal, D. and Kricitober, J.D. and Leijser, L. and LePine, M. and Lim, Y.-P. and Lodha, A. and Londhe, A. and Ly, L. and Maes, E. and Malhotra, A. and Marlow, N. and Mathew, J.L. and McDonald, C. and McLean, M. and Metcalfe, C. and Meyer, R. and Miller, S.P. and Miller, S. and Mogajane, T. and Mohammad, K. and Momin, S. and Montpetit, J. and Mukiza, N. and Murthy, P. and Scott, J.N. and Nakibuuka, V. and Nakwa, F. and Naulaers, G. and Noort, J. and Ntuli, N. and Ondongo-Ezhet, C. and Paul, R. and Pepper, M. and Plum, A. and Rombough, B. and Saugstad, O. and Scotland, J. and Scott, J. and Seake, K. and Sebunya, R. and Selvanathan, T. and Sepeng, L. and Simsek, H. and Steins-Rang, C. and Stonestreet, B. and Tang, S. and Taskin, E. and Thewissen, L. and Thomas, S. and Thomas, R. and van Kwawegen, A. and van Rensburg, J. and Velaphi, S. and Wu, Y. and Yaman, A. and Yapicioglu-Yildizdas, H. and Yawno, T. and Zaki, P. and Zein, H. and Zhou, L.
DOI: 10.3233/NPM-229003Children
Chan, N.H.-M. and Synnes, A. and Grunau, R.E. and Colby, L. and Petrie, J. and Elfring, T. and Richter, L. and Hendson, L. and Banihani, R. and Luu, T.M.
DOI: 10.3390/children9071048Journal of Pediatrics
Mclean, M.A. and Scoten, O.C. and Yu, W. and Ye, X.Y. and Petrie, J. and Church, P.T. and Soraisham, A.S. and Mirea, L.S. and Weinberg, J. and Synnes, A.R. and O{'}Brien, K. and Grunau, R.E.
DOI: 10.1016/j.jpeds.2021.12.055Archives of Disease in Childhood: Fetal and Neonatal Edition
Synnes, A.R. and Petrie, J. and Grunau, R.E. and Church, P. and Kelly, E. and Moddemann, D. and Ye, X. and Lee, S.K. and O{'}Brien, K.
DOI: 10.1136/archdischild-2020-321055Early Human Development
Duerden, E.G. and Mclean, M.A. and Chau, C. and Guo, T. and Mackay, M. and Chau, V. and Synnes, A. and Miller, S.P. and Grunau, R.E.
DOI: 10.1016/j.earlhumdev.2022.105617Biological Psychiatry
Miller, J.V. and Chau, V. and Synnes, A. and Miller, S.P. and Grunau, R.E.
DOI: 10.1016/j.biopsych.2022.03.010JAMA Network Open
Gomaa, N. and Konwar, C. and Gladish, N. and Au-Young, S.H. and Guo, T. and Sheng, M. and Merrill, S.M. and Kelly, E. and Chau, V. and Branson, H.M. and Ly, L.G. and Duerden, E.G. and Grunau, R.E. and Kobor, M.S. and Miller, S.P.
DOI: 10.1001/jamanetworkopen.2022.39796Scientific Reports
Wagner, M.W. and So, D. and Guo, T. and Erdman, L. and Sheng, M. and Ufkes, S. and Grunau, R.E. and Synnes, A. and Branson, H.M. and Chau, V. and Shroff, M.M. and Ertl-Wagner, B.B. and Miller, S.P.
DOI: 10.1038/s41598-022-16066-wJournal of Pediatrics
Sheng, M. and Guo, T. and Mabbott, C. and Chau, V. and Synnes, A. and de Vries, L.S. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2022.05.003Developmental Psychobiology
Mia A. McLean and Nikoo Niknafs and Olivia C. Scoten and Cecil M. Y. Chau and Margot MacKay and Joanne Weinberg and Anne Synnes and Steven P. Miller and Ruth E. Grunau
DOI: 10.1002/dev.2207907 / 2021
Pediatric Research
Christensen, R. and Chau, V. and Synnes, A. and Grunau, R.E. and Miller, S.P.
DOI: 10.1038/s41390-020-0840-7Prenatal Stress and Child Development
Levine, T.A. and Grunau, R.E.
DOI: 10.1007/978-3-030-60159-1_11Early Human Development
Ludwig, R.J. and Grunau, R.E. and Chafkin, J.E. and Hane, A.A. and Isler, J.R. and Chau, C.M.Y. and Welch, M.G. and Myers, M.
DOI: 10.1016/j.earlhumdev.2021.105455American Journal of Obstetrics and Gynecology
Roychoudhury, S. and Lodha, A. and Synnes, A. and Abou Mehrem, A. and Canning, R. and Banihani, R. and Beltempo, M. and Theriault, K. and Yang, J. and Shah, P.S. and Soraisham, A.S. and Ting, J. and Alvaro, R. and Adie, M. and Ng, E. and Pelausa, E. and Claveau, M. and Barrington, K. and Lapoint, A. and Ethier, G. and Drolet, C. and Piedboeuf, B. and Afifi, J. and Dahlgren, L. and Wood, S. and Metcalfe, A. and O'Quinn, C. and Helewa, M. and Taboun, F. and Melamed, N. and Abenhaim, H. and Wou, K. and Gratton, R. and Boucoiran, I. and Taillefer, C. and Allen, V. and Grunau, R. and Hendson, L. and Moddemann, D. and de Cabo, C. and Nwaesei, C. and Church, P. and Nguyen, K.-A. and Khairy, M. and Dorval, V. and Luu, T.M. and Blanger, S.
DOI: 10.1016/j.ajog.2021.03.027Annals of Neurology
Cayam-Rand, D. and Guo, T. and Synnes, A. and Chau, V. and Mabbott, C. and Benavente-Fern{\'a}ndez, I. and Grunau, R.E. and Miller, S.P.
DOI: 10.1002/ana.26201Developmental Psychobiology
Campbell, K.S.J. and Williams, L.J. and Bjornson, B.H. and Weik, E. and Brain, U. and Grunau, R.E. and Miller, S.P. and Oberlander, T.F.
DOI: 10.1002/dev.22125Paediatric and Neonatal Pain
Ruth Eckstein Grunau
DOI: 10.1002/pne2.1201706 / 2020
BJPsych Open
Dhaliwal, G. and Weikum, W.M. and Jolicoeur-Martineau, A. and Brain, U. and Grunau, R.E. and Oberlander, T.F.
DOI: 10.1192/bjo.2020.73Seminars in Fetal and Neonatal Medicine
Synnes, A. and Grunau, R.E.
DOI: 10.1016/j.siny.2020.101160Journal of Pediatrics
Guillot, M. and Guo, T. and Ufkes, S. and Schneider, J. and Synnes, A. and Chau, V. and Grunau, R.E. and Miller, S.P.
DOI: 10.1016/j.jpeds.2020.05.039Annals of Neurology
Garfinkle, J. and Guo, T. and Synnes, A. and Chau, V. and Branson, H.M. and Ufkes, S. and Tam, E.W.Y. and Grunau, R.E. and Miller, S.P.
DOI: 10.1002/ana.25899Neurology
Duerden, E.G. and Grunau, R.E. and Chau, V. and Groenendaal, F. and Guo, T. and Chakravarty, M.M. and Benders, M. and Wagenaar, N. and Eijsermans, R. and Koopman, C. and Synnes, A. and Vries, L. and Miller, S.P.
DOI: 10.1212/WNL.0000000000010953Human Brain Mapping
Kozhemiako, N. and Nunes, A.S. and Vakorin, V.A. and Chau, C.M.Y. and Moiseev, A. and Ribary, U. and Grunau, R.E. and Doesburg, S.M.
DOI: 10.1002/hbm.24809Early Human Development
Church, P.T. and Grunau, R.E. and Mirea, L. and Petrie, J. and Soraisham, A.S. and Synnes, A. and Ye, X.Y. and O{'}Brien, K.
DOI: 10.1016/j.earlhumdev.2020.105196Adonay S. Nunes and Nataliia Kozhemiako and Evan Hutcheon and Cecil Chau and Urs Ribary and Ruth E Grunau and Sam M Doesburg
DOI: 10.1101/72903808 / 2019
Canadian Journal of Pain
Carl L. von Baeyer and Bonnie J. Stevens and Kenneth D. Craig and G. Allen Finley and C. Celeste Johnston and Ruth V.E. Grunau and Christine T. Chambers and Rebecca R. Pillai Riddell and Jennifer N. Stinson and Patrick J. McGrath
DOI: 10.1080/24740527.2018.156284401 / 2019
Journal of Perinatology
Levine, T.A. and Grunau, R.E. and McAuliffe, F.M. and Alderdice, F.A.
DOI: 10.1038/s41372-019-0369-yEBioMedicine
Chau, C.M.Y. and Ross, C.J.D. and Chau, V. and Synnes, A.R. and Miller, S.P. and Carleton, B. and Grunau, R.E.
DOI: 10.1016/j.ebiom.2019.01.042Journal of Child Psychology and Psychiatry and Allied Disciplines
Kozhemiako, N. and Nunes, A. and Vakorin, V.A. and Chau, C.M.Y. and Moiseev, A. and Ribary, U. and Grunau, R.E. and Doesburg, S.M.
DOI: 10.1111/jcpp.13026Journal of Affective Disorders
Erickson, N.L. and Hancock, G.R. and Oberlander, T.F. and Brain, U. and Grunau, R.E. and Gartstein, M.A.
DOI: 10.1016/j.jad.2019.08.003Computerized Medical Imaging and Graphics
Brown, C.J. and Miller, S.P. and Booth, B.G. and Zwicker, J.G. and Grunau, R.E. and Synnes, A.R. and Chau, V. and Hamarneh, G.
DOI: 10.1016/j.compmedimag.2018.08.009Journal of Child Neurology
Tam, E.W.Y. and Chau, V. and Lavoie, R. and Chakravarty, M.M. and Guo, T. and Synnes, A. and Zwicker, J. and Grunau, R. and Miller, S.P.
DOI: 10.1177/0883073819847925Frontiers in Behavioral Neuroscience
Chau, C.M.Y. and Ranger, M. and Bichin, M. and Park, M.T.M. and Amaral, R.S.C. and Chakravarty, M. and Poskitt, K. and Synnes, A.R. and Miller, S.P. and Grunau, R.E.
DOI: 10.3389/fnbeh.2019.00051JAMA Pediatrics
Schmidt, B. and Anderson, P.J. and Asztalos, E.V. and Doyle, L.W. and Grunau, R.E. and Moddemann, D. and Roberts, R.S.
DOI: 10.1001/jamapediatrics.2018.4853JAMA Network Open
Benavente-Fernandez, I. and Synnes, A. and Grunau, R.E. and Chau, V. and Ramraj, C. and Glass, T. and Cayam-Rand, D. and Siddiqi, A. and Miller, S.P.
DOI: 10.1001/jamanetworkopen.2019.2914Biological Psychiatry: Cognitive Neuroscience and Neuroimaging
Rotem-Kohavi, N. and Williams, L.J. and Virji-Babul, N. and Bjornson, B.H. and Brain, U. and Werker, J.F. and Grunau, R.E. and Miller, S.P. and Oberlander, T.F.
DOI: 10.1016/j.bpsc.2018.08.004Physical and Occupational Therapy in Pediatrics
Gill, K. and Osiovich, A. and Synnes, A. and A. Agnew, J. and Grunau, R.E. and Miller, S.P. and Zwicker, J.G.
DOI: 10.1080/01942638.2018.1546255Neurology
Cayam-Rand, D. and Guo, T. and Grunau, R.E. and Benavente-Fern{\'a}ndez, I. and Synnes, A. and Chau, V. and Branson, H. and Latal, B. and Mcquillen, P. and Miller, S.P.
DOI: 10.1212/WNL.0000000000008172Depression and Anxiety
Rotem-Kohavi, N. and Williams, L.J. and Muller, A.M. and Abdi, H. and Virji-Babul, N. and Bjornson, B.H. and Brain, U. and Werker, J.F. and Grunau, R.E. and Miller, S.P. and Oberlander, T.F.
DOI: 10.1002/da.22906Frontiers in Psychology
Ranger, M. and Tremblay, S. and Chau, C.M.Y. and Holsti, L. and Grunau, R.E. and Goldowitz, D.
DOI: 10.3389/fpsyg.2018.02394Archives of Women's Mental Health
Park, M. and Brain, U. and Grunau, R.E. and Diamond, A. and Oberlander, T.F.
DOI: 10.1007/s00737-017-0803-0Pain
Schneider, J. and Duerden, E.G. and Guo, T. and Ng, K. and Hagmann, P. and Graz, M.B. and Grunau, R.E. and Chakravarty, M.M. and Huppi, P.S. and Truttmann, A.C. and Miller, S.P.
DOI: 10.1097/j.pain.0000000000001123Journal of Neuroscience
Duerden, E.G. and Grunau, R.E. and Guo, T. and Foong, J. and Pearson, A. and Au-Young, S. and Lavoie, R. and Chakravarty, M.M. and Chau, V. and Synnes, A. and Miller, S.P.
DOI: 10.1523/JNEUROSCI.0867-17.2017Development and Psychopathology
Neuenschwander, R. and Hookenson, K. and Brain, U. and Grunau, R.E. and Devlin, A.M. and Weinberg, J. and Diamond, A. and Oberlander, T.F.
DOI: 10.1017/S095457941800041XPediatrics
M{\"u}rner-Lavanchy, I.M. and Doyle, L.W. and Schmidt, B. and Roberts, R.S. and Asztalos, E.V. and Costantini, L. and Davis, P.G. and Dewey, D. and D{'}Ilario, J. and Grunau, R.E. and Moddemann, D. and Nelson, H. and Ohlsson, A. and Solimano, A. and Tin, W. and Anderson, P.J. and Dix, J. and Adams, B.A. and Warriner, E. and Callanan, C. and Davis, N. and McDonald, M. and Duff, J. and Kelly, E. and Hutchinson, E. and Hohn, D. and Ayaz, A. and Allen, J. and Haslam, R. and Goodchild, L. and Lontis, R.M. and Opie, G. and Woods, H. and Marchant, E. and Magrath, E. and Williamson, A. and Synnes, A. and Butt, A. and Petrie, J. and Sauve, R.S. and Christianson, H. and Anseeuw-Deeks, D. and Makarchuk, S. and Debooy, V. and Granke, N. and Bow, J. and Herlenius, E. and Legnevall, L. and B{\"o}hm, B. and BergStr{\"o}m, B.-M. and St?lnacke, S. and Sund{\'e}n-Cullberg, S. and Mayes, C. and McCusker, C. and Robinson, U. and Embleton, N. and Carnell, J.
DOI: 10.1542/peds.2017-4047JAMA Pediatrics
Bierstone, D. and Wagenaar, N. and Gano, D.L. and Guo, T. and Georgio, G. and Groenendaal, F. and De Vries, L.S. and Varghese, J. and Glass, H.C. and Chung, C. and Terry, J. and Rijpert, M. and Grunau, R.E. and Synnes, A. and Barkovich, A.J. and Ferriero, D.M. and Benders, M. and Chau, V. and Miller, S.P.
DOI: 10.1001/jamapediatrics.2018.0102Journal of Nutrition
Wiedeman, A.M. and Chau, C.M.Y. and Grunau, R.E. and McCarthy, D. and Yurko-Mauro, K. and Dyer, R.A. and Innis, S.M. and Devlin, A.M.
DOI: 10.1093/jn/nxy108Journal of Pediatrics
Glass, T.J.A. and Chau, V. and Grunau, R.E. and Synnes, A. and Guo, T. and Duerden, E.G. and Foong, J. and Poskitt, K.J. and Miller, S.P.
DOI: 10.1016/j.jpeds.2017.12.041Journal of Pediatrics
Wagenaar, N. and Chau, V. and Groenendaal, F. and Kersbergen, K.J. and Poskitt, K.J. and Grunau, R.E. and Synnes, A. and Duerden, E.G. and de Vries, L.S. and Miller, S.P. and Benders, M.J.N.L.
DOI: 10.1016/j.jpeds.2016.11.073Pain
Tremblay, S. and Ranger, M. and Chau, C.M.Y. and Ellegood, J. and Lerch, J.P. and Holsti, L. and Goldowitz, D. and Grunau, R.E.
DOI: 10.1097/j.pain.0000000000000961Neurology
Guo, T. and Duerden, E.G. and Adams, E. and Chau, V. and Branson, H.M. and Chakravarty, M.M. and Poskitt, K.J. and Synnes, A. and Grunau, R.E. and Miller, S.P.
DOI: 10.1212/WNL.0000000000003606American Journal of Perinatology
Bashir, R.A. and Thomas, J.P. and Mackay, M. and Agnew, J. and Hubber-Richard, P. and Grunau, R.E. and Synnes, A.
DOI: 10.1055/s-0037-1603462Journal of Pediatrics
Borenstein-Levin, L. and Synnes, A. and Grunau, R.E. and Miller, S.P. and Yoon, E.W. and Shah, P.S.
DOI: 10.1016/j.jpeds.2016.08.031NeuroImage
Kawahara, J. and Brown, C.J. and Miller, S.P. and Booth, B.G. and Chau, V. and Grunau, R.E. and Zwicker, J.G. and Hamarneh, G.
DOI: 10.1016/j.neuroimage.2016.09.046BMJ Open
Levine, T.A. and Grunau, R.E. and Segurado, R. and Daly, S. and Geary, M.P. and Kennelly, M.M. and O{'}Donoghue, K. and Hunter, A. and Morrison, J.J. and Burke, G. and Dicker, P. and Tully, E.C. and Malone, F.D. and Alderdice, F.A. and McAuliffe, F.M.
DOI: 10.1136/bmjopen-2016-015326JAMA Pediatrics
Schmidt, B. and Roberts, R.S. and Anderson, P.J. and Asztalos, E.V. and Costantini, L. and Davis, P.G. and Dewey, D. and D{'}Ilario, J. and Doyle, L.W. and Grunau, R.E. and Moddemann, D. and Nelson, H. and Ohlsson, A. and Solimano, A. and Tin, W. and Dix, J. and Adams, B.A. and Warriner, E. and Callanan, C. and Davis, N. and McDonald, M. and Duff, J. and Kelly, E. and Hutchinson, E. and Hohn, D. and Ayaz, A. and Allen, J. and Haslam, R. and Goodchild, L. and Lontis, R.M. and Opie, G. and Woods, H. and Marchant, E. and Magrath, E. and Williamson, A. and Bairam, A. and B{\'e}langer, S. and Fraser, A. and Leblanc, M. and Synnes, A. and Butt, A. and Petrie, J. and Sauve, R.S. and Christianson, H. and Anseeuw-Deeks, D. and Makarchuk, S. and Debooy, V. and Granke, N. and Bow, J. and Nwaesei, C. and Ryan, H. and Saunders, C. and Herlenius, E. and Legnevall, L. and B{\"o}hm, B. and Bergstr{\"o}m, B.-M. and St?lnacke, S. and Sund{\'e}n-Cullberg, S. and Mayes, C. and McCusker, C. and Robinson, U. and Embleton, N. and Carnell, J.
DOI: 10.1001/jamapediatrics.2017.0238Pain Medicine (United States)
Shavit, I. and Peri-Front, Y. and Rosen-Walther, A. and Grunau, R.E. and Neuman, G. and Nachmani, O. and Koren, G. and Aizenbud, D.
DOI: 10.1093/pm/pnw097Neuroscience
Chau, C.M.Y. and Cepeda, I.L. and Devlin, A.M. and Weinberg, J. and Grunau, R.E.
DOI: 10.1016/j.neuroscience.2015.08.044Fetal and Neonatal Physiology, 2-Volume Set
Ranger, M. and Beggs, S. and Grunau, R.E.
DOI: 10.1016/B978-0-323-35214-7.00137-2Neuroscience
Podrebarac, S.K. and Duerden, E.G. and Chau, V. and Grunau, R.E. and Synnes, A. and Oberlander, T.F. and Miller, S.P.
DOI: 10.1016/j.neuroscience.2016.11.025Archives of Disease in Childhood: Fetal and Neonatal Edition
Glass, T.J.A. and Chau, V. and Gardiner, J. and Foong, J. and Vinall, J. and Zwicker, J.G. and Grunau, R.E. and Synnes, A. and Poskitt, K.J. and Miller, S.P.
DOI: 10.1136/archdischild-2016-312533Developmental Medicine and Child Neurology
Malavolti, A.M. and Chau, V. and Brown-Lum, M. and Poskitt, K.J. and Brant, R. and Synnes, A. and Grunau, R.E. and Miller, S.P.
DOI: 10.1111/dmcn.13364Nutrients
Devlin, A.M. and Chau, C.M.Y. and Dyer, R. and Matheson, J. and McCarthy, D. and Yurko-Mauro, K. and Innis, S.M. and Grunau, R.E.
DOI: 10.3390/nu9090975Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics)
Brown, C.J. and Moriarty, K.P. and Miller, S.P. and Booth, B.G. and Zwicker, J.G. and Grunau, R.E. and Synnes, A.R. and Chau, V. and Hamarneh, G.
DOI: 10.1007/978-3-319-66182-7_10Archives of Women's Mental Health
Levine, T.A. and Alderdice, F.A. and Grunau, R.E. and McAuliffe, F.M.
DOI: 10.1007/s00737-016-0645-1Clinical Journal of Pain
Valeri, B.O. and Ranger, M. and Chau, C.M.Y. and Cepeda, I.L. and Synnes, A. and Linhares, M.B.M. and Grunau, R.E.
DOI: 10.1097/AJP.0000000000000353Epigenomics
Montirosso, R. and Provenzi, L. and Giorda, R. and Fumagalli, M. and Morandi, F. and Sirgiovanni, I. and Pozzoli, U. and Grunau, R. and Oberlander, T.F. and Mosca, F. and Borgatti, R.
DOI: 10.2217/epi-2016-0010NeuroImage
Booth, B.G. and Miller, S.P. and Brown, C.J. and Poskitt, K.J. and Chau, V. and Grunau, R.E. and Synnes, A.R. and Hamarneh, G.
DOI: 10.1016/j.neuroimage.2015.08.079Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics)
Brown, C.J. and Miller, S.P. and Booth, B.G. and Zwicker, J.G. and Grunau, R.E. and Synnes, A.R. and Chau, V. and Hamarneh, G.
DOI: 10.1007/978-3-319-46720-7_21Neonatology
Holsti, L. and Zwicker, J.G. and Grunau, R.E.
DOI: 10.1159/000442081Annals of Neurology
Duerden, E.G. and Guo, T. and Dodbiba, L. and Chakravarty, M.M. and Chau, V. and Poskitt, K.J. and Synnes, A. and Grunau, R.E. and Miller, S.P.
DOI: 10.1002/ana.24601Hypertension
Hovi, P. and Vohr, B. and Ment, L.R. and Doyle, L.W. and McGarvey, L. and Morrison, K.M. and Evensen, K.A.I. and Van Der Pal, S. and Grunau, R.E. and Brubakk, A.-M. and Andersson, S. and Saigal, S. and Kajantie, E.
DOI: 10.1161/HYPERTENSIONAHA.116.08167Journal of Pediatrics
Zwicker, J.G. and Miller, S.P. and Grunau, R.E. and Chau, V. and Brant, R. and Studholme, C. and Liu, M. and Synnes, A. and Poskitt, K.J. and Stiver, M.L. and Tam, E.W.Y.
DOI: 10.1016/j.jpeds.2015.12.024Developmental Psychobiology
Gartstein, M.A. and Hookenson, K.V. and Brain, U. and Devlin, A.M. and Grunau, R.E. and Oberlander, T.F.
DOI: 10.1002/dev.21414Child Neuropsychology
Wagner, S.L. and Cepeda, I. and Krieger, D. and Maggi, S. and D?Angiulli, A. and Weinberg, J. and Grunau, R.E.
DOI: 10.1080/09297049.2015.1080232Pediatric Research
Tam, E.W.Y. and Chau, V. and Barkovich, A.J. and Ferriero, D.M. and Miller, S.P. and Rogers, E.E. and Grunau, R.E. and Synnes, A.R. and Xu, D. and Foong, J. and Brant, R. and Innis, S.M.
DOI: 10.1038/pr.2016.11Progress in Biomedical Optics and Imaging - Proceedings of SPIE
Garg, A. and Grunau, R.E. and Popuri, K. and Miller, S. and Bjornson, B. and Poskitt, K.J. and Beg, M.F.
DOI: 10.1117/12.2216914American Journal of Occupational Therapy
Crozier, S.C. and Goodson, J.Z. and Mackay, M.L. and Synnes, A.R. and Grunau, R.E. and Miller, S.P. and Zwicker, J.G.
DOI: 10.5014/ajot.2016.018747Brain Topography
Moiseev, A. and Doesburg, S.M. and Herdman, A.T. and Ribary, U. and Grunau, R.E.
DOI: 10.1007/s10548-014-0416-0Archives of Disease in Childhood
Synnes, A. and Anderson, P.J. and Grunau, R.E. and Dewey, D. and Moddemann, D. and Tin, W. and Davis, P.G. and Doyle, L.W. and Foster, G. and Khairy, M. and Nwaesei, C. and Schmidt, B. and D{'}Ilario, J. and Cairnie, J. and Dix, J. and Adams, B.A. and Warriner, E. and Kim, M.-H.M. and Argus, B. and Callanan, C. and Davis, N. and Duff, J. and McDonald, M. and Asztalos, E. and Hohn, D. and Lacy, M. and Haslam, R. and Barnett, C. and Goodchild, L. and Lontis, R.M. and Fraser, S. and Keng, J. and Saunders, K. and Opie, G. and Kelly, E. and Woods, H. and Marchant, E. and Turner, A.-M. and Magrath, E. and Williamson, A. and Bairam, A. and B{\'e}langer, S. and Fraser, A. and Blayney, M. and Lemyre, B. and Frank, J. and Solimano, A. and Hubber-Richard, P. and Rogers, M. and Mackay, M. and Petrie-Thomas, J. and Butt, A. and Van Wassenaer, A. and Nuytemans, D. and Houtzager, B. and Van Sonderen, L. and Regev, R. and Itzchack, N. and Arnon, S. and Chalaf, A. and Ohlsson, A. and O{'}Brien, K. and Hamilton, A.-M. and Chan, M.L. and Sankaran, K. and Proctor, P. and Golan, A. and Goldsch-Lerman, E. and Reynolds, G. and Dromgool, B. and Meskell, A. and Parr, V. and Maher, C. and Broom, M. and Kecskes, Z. and Ringland, C. and McMillan, D. and Spellen, E. and Sauve, R.S. and Christianson, H. and Anseeuw-Deeks, D. and Creighton, D. and Heath, J. and Alvaro, R. and Chiu, A. and Porter, C. and Turner, G. and Granke, N. and Penner, K. and Bow, J. and Mulder, A. and Wassenberg, R. and Van Der Hoeven, M. and Clarke, M. and Parfitt, J. and Parker, K. and Ryan, H. and Saunders, C. and Schulze, A. and Wermuth, I. and Hilgendorff, A. and Flemmer, A.W. and Herlenius, E. and Legnevall, L. and Lagercrantz, H. and Matthew, D. and Amos, W. and Tulsiani, S. and Tan-Dy, C. and Turner, M. and Phelan, C. and Shinwell, E.S. and Levine, M. and Juster-Reicher, A. and Grier, P. and Vachon, J. and Perepolkin, L. and Barrington, K.J. and Sinha, S.K. and Fritz, S. and Walti, H. and Royer, D. and Halliday, H. and Millar, D. and Mayes, C. and McCusker, C. and McLaughlin, O. and Fahnenstich, H. and Tillmann, B. and Weber, P. and Wariyar, U. and Embleton, N. and Swamy, R. and Bucher, H.U. and Fauchere, J.-C. and Dietz, V. and Harikumar, C.
DOI: 10.1136/archdischild-2014-307695NeuroImage
Moiseev, A. and Doesburg, S.M. and Grunau, R.E. and Ribary, U.
DOI: 10.1016/j.neuroimage.2015.06.079Early Human Development
Perra, O. and McGowan, J.E. and Grunau, R.E. and Doran, J.B. and Craig, S. and Johnston, L. and Jenkins, J. and Holmes, V.A. and Alderdice, F.A.
DOI: 10.1016/j.earlhumdev.2015.01.009Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics)
Brown, C.J. and Miller, S.P. and Booth, B.G. and Poskitt, K.J. and Chau, V. and Synnes, A.R. and Zwicker, J.G. and Grunau, R.E. and Hamarneh, G.
DOI: 10.1007/978-3-319-24553-9_9Pediatrics
Levine, T.A. and Grunau, R.E. and McAuliffe, F.M. and Pinnamaneni, R.M. and Foran, A. and Alderdice, F.A.
DOI: 10.1542/peds.2014-1143Journal of Pediatrics
Ranger, M. and Zwicker, J.G. and Chau, C.M.Y. and Park, M.T.M. and Chakravarthy, M.M. and Poskitt, K. and Miller, S.P. and Bjornson, B.H. and Tam, E.W.Y. and Chau, V. and Synnes, A.R. and Grunau, R.E.
DOI: 10.1016/j.jpeds.2015.04.055American Journal of Neuroradiology
Duerden, E.G. and Foong, J. and Chau, V. and Branson, H. and Poskitt, K.J. and Grunau, R.E. and Synnes, A. and Zwicker, J.G. and Miller, S.P.
DOI: 10.3174/ajnr.A4312Archives of Disease in Childhood
Linden, M.A. and Cepeda, I.L. and Synnes, A. and Grunau, R.E.
DOI: 10.1136/archdischild-2014-307390Journal of Pediatrics
Manley, B.J. and Roberts, R.S. and Doyle, L.W. and Schmidt, B. and Anderson, P.J. and Barrington, K.J. and B{\"o}hm, B. and Golan, A. and Van Wassenaer-Leemhuis, A.G. and Davis, P.G. and D{'}Ilario, J. and Cairnie, J. and Dix, J. and Adams, B.A. and Warriner, E. and Kim, M.-H.M. and Argus, B. and Callanan, C. and Duff, J. and McDonald, M. and Asztalos, E. and Hohn, D. and Lacy, M. and Haslam, R. and Barnett, C. and Goodchild, L. and Lontis, R.M. and Fraser, S. and Keng, J. and Saunders, K. and Opie, G. and Kelly, E. and Woods, H. and Marchant, E. and Turner, A.-M. and Davis, N. and Magrath, E. and Williamson, A. and Bairam, A. and Belanger, S. and Fraser, A. and Blayney, M. and Lemyre, B. and Frank, J. and Solimano, A. and Synnes, A. and Grunau, R.E. and Hubber-Richard, P. and Rogers, M. and Mackay, M. and Petrie-Thomas, J. and Butt, A. and Van Wassenaer, A. and Nuytemans, D. and Houtzager, B. and Van Sonderen, L. and Regev, R. and Itzchack, N. and Arnon, S. and Chalaf, A. and Ohlsson, A. and O{'}Brien, K. and Hamilton, A.-M. and Chan, M.L. and Sankaran, K. and Proctor, P. and Goldsch-Lerman, E. and Reynolds, G. and Dromgool, B. and Meskell, S. and Parr, V. and Maher, C. and Broom, M. and Kecskes, Z. and Ringland, C. and McMillan, D. and Spellen, E. and Sauve, R.S. and Christianson, H. and Anseeuw-Deeks, D. and Creighton, D. and Heath, J. and Alvaro, R. and Chiu, A. and Porter, C. and Turner, G. and Moddemann, D. and Granke, N. and Penner, K. and Bow, J. and Mulder, A. and Wassenberg, R. and Van Der Hoeven, M. and Clarke, M. and Parfitt, J. and Parker, K. and Nwaesei, C. and Ryan, H. and Saunders, C. and Schulze, A. and Wermuth, I. and Hilgendorff, A. and Flemmer, A.W. and Herlenius, E. and Legnevall, L. and Lagercrantz, H. and Matthew, D. and Amos, W. and Tulsiani, S. and Tan-Dy, C. and Turner, M. and Phelan, C. and Shinwell, E.S. and Levine, M. and Juster-Reicher, A. and Khairy, M. and Grier, P. and Vachon, J. and Perepolkin, L. and Sinha, S.K. and Tin, W. and Fritz, S. and Walti, H. and Royer, D. and Halliday, H. and Millar, D. and Mayes, C. and McCusker, C. and McLaughlin, O. and Fahnenstich, H. and Tillmann, B. and Weber, P. and Wariyar, U. and Embleton, N. and Swamy, R. and Bucher, H.U. and Fauchere, J.-C. and Dietz, V. and Harikumar, C. and Dewey, D. and Gent, M. and Fraser, W. and Hey, E. and Perlman, M. and Thorpe, K. and Gray, S. and Chambers, C. and Costantini, L. and Yacura, W. and McGean, E. and Scapinello, L.
DOI: 10.1016/j.jpeds.2014.12.016NeoReviews
Cheung, T.P.L. and Grunau, R.E. and Synnes, A. and Eswaran, H. and Doesburg, S.M.
DOI: 10.1542/neo.16-9-e544NeuroImage: Clinical
Guo, T. and Winterburn, J.L. and Pipitone, J. and Duerden, E.G. and Park, M.T.M. and Chau, V. and Poskitt, K.J. and Grunau, R.E. and Synnes, A. and Miller, S.P. and Mallar Chakravarty, M.
DOI: 10.1016/j.nicl.2015.07.019Psychoneuroendocrinology
Brummelte, S. and Chau, C.M.Y. and Cepeda, I.L. and Degenhardt, A. and Weinberg, J. and Synnes, A.R. and Grunau, R.E.
DOI: 10.1016/j.psyneuen.2014.09.018European Journal of Pain (United Kingdom)
Ranger, M. and Synnes, A.R. and Vinall, J. and Grunau, R.E.
DOI: 10.1002/j.1532-2149.2013.00431.xFrontiers in Behavioral Neuroscience
Chau, C.M.Y. and Ranger, M. and Sulistyoningrum, D. and Devlin, A.M. and Obertander, T.F. and Grunau, R.E.
DOI: 10.3389/fnbeh.2014.00409Clinics in Perinatology
McPherson, C. and Grunau, R.E.
DOI: 10.1016/j.clp.2013.10.002Journal of Pediatrics
Doyle, L.W. and Schmidt, B. and Anderson, P.J. and Davis, P.G. and Moddemann, D. and Grunau, R.E. and O{'}Brien, K. and Sankaran, K. and Herlenius, E. and Roberts, R.
DOI: 10.1016/j.jpeds.2014.04.016Journal of Pediatrics
De Graaf, J. and Van Den Akker, E.L.T. and Van Lingen, R.A. and Groot Jebbink, L.J.M. and De Jong, F.H. and Grunau, R.E. and Van Dijk, M. and Tibboel, D.
DOI: 10.1016/j.jpeds.2014.05.047Pediatric Research
Vinall, J. and Grunau, R.E.
DOI: 10.1038/pr.2014.16Pain Research and Management
Von Baeyer, C.L. and Stevens, B.J. and Chambers, C.T. and Craig, K.D. and Finley, G.A. and Grunau, R.E. and Johnston, C.C. and Riddell, R.P. and Stinson, J.N. and Dol, J. and Campbell-Yeo, M. and McGrath, P.J.
DOI: 10.1155/2014/692857Pain management
Ranger, M. and Grunau, R.E.
DOI: 10.2217/pmt.13.61Journal of Medical Imaging
Garg, A. and Wong, D. and Popuri, K. and Poskitt, K.J. and Fitzpatrick, K. and Bjornson, B. and Grunau, R.E. and Beg, M.F.
DOI: 10.1117/1.JMI.1.3.034502Pediatrics
Vinall, J. and Miller, S.P. and Bjornson, B.H. and Fitzpatrick, K.P.V. and Poskitt, K.J. and Brant, R. and Synnes, A.R. and Cepeda, I.L. and Grunau, R.E.
DOI: 10.1542/peds.2013-1863Frontiers in Cellular Neuroscience
Weikum, W.M. and Brain, U. and Chau, C.M.Y. and Grunau, R.E. and Boyce, W.T. and Diamond, A. and Oberlander, T.F.
DOI: 10.3389/fncel.2013.00180Infant Behavior and Development
Weikum, W.M. and Mayes, L.C. and Grunau, R.E. and Brain, U. and Oberlander, T.F.
DOI: 10.1016/j.infbeh.2013.04.001Child and Youth Care Forum
Wagner, S.L. and Forer, B. and Cepeda, I.L. and Goelman, H. and Maggi, S. and D{'}Angiulli, A. and Wessel, J. and Hertzman, C. and Grunau, R.E.
DOI: 10.1007/s10566-012-9187-5Pediatric Neurology
Zwicker, J.G. and Grunau, R.E. and Adams, E. and Chau, V. and Brant, R. and Poskitt, K.J. and Synnes, A. and Miller, S.P.
DOI: 10.1016/j.pediatrneurol.2012.10.016Pain
Doesburg, S.M. and Chau, C.M. and Cheung, T.P.L. and Moiseev, A. and Ribary, U. and Herdman, A.T. and Miller, S.P. and Cepeda, I.L. and Synnes, A. and Grunau, R.E.
DOI: 10.1016/j.pain.2013.04.009PLoS ONE
Ranger, M. and Chau, C.M.Y. and Garg, A. and Woodward, T.S. and Beg, M.F. and Bjornson, B. and Poskitt, K. and Fitzpatrick, K. and Synnes, A.R. and Miller, S.P. and Grunau, R.E.
DOI: 10.1371/journal.pone.0076702Neuroradiology
Duerden, E.G. and Brown-Lum, M. and Chau, V. and Poskitt, K.J. and Grunau, R.E. and Synnes, A. and Miller, S.P.
DOI: 10.1007/s00234-013-1243-9Frontiers in Human Neuroscience
Doesburg, S.M. and Moiseev, A. and Herdman, A.T. and Ribary, U. and Grunau, R.E.
DOI: 10.3389/fnhum.2013.00791Journal of Perinatology
Gover, A. and Chau, V. and Miller, S.P. and Brant, R. and Mcfadden, D.E. and Poskitt, K.J. and Synnes, A. and Weinberg, J. and Grunau, R.E.
DOI: 10.1038/jp.2013.24PLoS ONE
Grunau, R.E. and Cepeda, I.L. and Chau, C.M.Y. and Brummelte, S. and Weinberg, J. and Lavoie, P.M. and Ladd, M. and Hirschfeld, A.F. and Russell, E. and Koren, G. and Van Uum, S. and Brant, R. and Turvey, S.E.
DOI: 10.1371/journal.pone.0073926Enfance
Grunau, R.E.
DOI: 10.4074/S0013754513001043Pain
Vinall, J. and Miller, S.P. and Synnes, A.R. and Grunau, R.E.
DOI: 10.1016/j.pain.2013.05.050Psychoneuroendocrinology
Vaghri, Z. and Guhn, M. and Weinberg, J. and Grunau, R.E. and Yu, W. and Hertzman, C.
DOI: 10.1016/j.psyneuen.2012.06.009Clinical Journal of Pain
Maimon, N. and Grunau, R.E. and Cepeda, I.L. and Friger, M. and Selnovik, L. and Gilat, S. and Shany, E.
DOI: 10.1097/AJP.0b013e318284e525Neurology
Chau, V. and Synnes, A. and Grunau, R.E. and Poskitt, K.J. and Brant, R. and Miller, S.P.
DOI: 10.1212/01.wnl.0000437298.43688.b9Science Translational Medicine
Vinall, J. and Grunau, R.E. and Brant, R. and Chau, V. and Poskitt, K.J. and Synnes, A.R. and Miller, S.P.
DOI: 10.1126/scitranslmed.3004666Developmental Psychobiology
Petrie Thomas, J.H. and Whitfield, M.F. and Oberlander, T.F. and Synnes, A.R. and Grunau, R.E.
DOI: 10.1002/dev.20597JAMA - Journal of the American Medical Association
Schmidt, B. and Anderson, P.J. and Doyle, L.W. and Dewey, D. and Grunau, R.E. and Asztalos, E.V. and Davis, P.G. and Tin, W. and Moddemann, D. and Solimano, A. and Ohlsson, A. and Barrington, K.J. and Roberts, R.S.
DOI: 10.1001/jama.2011.2024Pain
Vinall, J. and Miller, S.P. and Chau, V. and Brummelte, S. and Synnes, A.R. and Grunau, R.E.
DOI: 10.1016/j.pain.2012.02.007Acta Paediatrica, International Journal of Paediatrics
Gover, A. and Brummelte, S. and Synnes, A.R. and Miller, S.P. and Brant, R. and Weinberg, J. and Grunau, R.E.
DOI: 10.1111/j.1651-2227.2012.02629.xAnnals of Neurology
Brummelte, S. and Grunau, R.E. and Chau, V. and Poskitt, K.J. and Brant, R. and Vinall, J. and Gover, A. and Synnes, A.R. and Miller, S.P.
DOI: 10.1002/ana.22267Frontiers in Human Neuroscience
D{'}Angiulli, A. and Van Roon, P.M. and Weinberg, J. and Oberlander, T.F. and Grunau, R.E. and Hertzman, C. and Maggi, S.
DOI: 10.3389/fnhum.2012.00306Pediatric Research
Doesburg, S.M. and Ribary, U. and Herdman, A.T. and Moiseev, A. and Cheung, T. and Miller, S.P. and Poskitt, K.J. and Weinberg, H. and Whitfield, M.F. and Synnes, A. and Grunau, R.E.
DOI: 10.1203/PDR.0b013e3182225a9eEarly Human Development
Brummelte, S. and Grunau, R.E. and Synnes, A.R. and Whitfield, M.F. and Petrie-Thomas, J.
DOI: 10.1016/j.earlhumdev.2011.01.030NeuroImage
Doesburg, S.M. and Ribary, U. and Herdman, A.T. and Miller, S.P. and Poskitt, K.J. and Moiseev, A. and Whitfield, M.F. and Synnes, A. and Grunau, R.E.
DOI: 10.1016/j.neuroimage.2010.10.044Developmental Psychobiology
Brummelte, S. and Grunau, R.E. and Zaidman-Zait, A. and Weinberg, J. and Nordstokke, D. and Cepeda, I.L.
DOI: 10.1002/dev.20511Science Translational Medicine
Tam, E.W.Y. and Chau, V. and Ferriero, D.M. and Barkovich, A.J. and Poskitt, K.J. and Studholme, C. and Fok, E.D.-Y. and Grunau, R.E. and Glidden, D.V. and Miller, S.P.
DOI: 10.1126/scitranslmed.3002884Clinical Journal of Pain
Grunau, R.E. and Tu, M.T. and Whitfield, M.F. and Oberlander, T.F. and Weinberg, J. and Yu, W. and Thiessen, P. and Gosse, G. and Scheifele, D.
DOI: 10.1097/AJP.0b013e3181e5bb00Pediatrics
Holsti, L. and Grunau, R.E.
DOI: 10.1542/peds.2009-2445Journal of Pediatrics
Adams, E. and Chau, V. and Poskitt, K.J. and Grunau, R.E. and Synnes, A. and Miller, S.P.
DOI: 10.1016/j.jpeds.2009.12.030Experimental Brain Research
Doesburg, S.M. and Herdman, A.T. and Ribary, U. and Cheung, T. and Moiseev, A. and Weinberg, H. and Liotti, M. and Weeks, D. and Grunau, R.E.
DOI: 10.1007/s00221-009-2086-9Journal of Pediatrics
Synnes, A.R. and Anson, S. and Arkesteijn, A. and Butt, A. and Grunau, R.E. and Rogers, M. and Whitfield, M.F.
DOI: 10.1016/j.jpeds.2010.06.016Alcoholism: Clinical and Experimental Research
Oberlander, T.F. and Jacobson, S.W. and Weinberg, J. and Grunau, R.E. and Molteno, C.D. and Jacobson, J.L.
DOI: 10.1111/j.1530-0277.2009.01137.xInfant Behavior and Development
Haley, D.W. and Grunau, R.E. and Weinberg, J. and Keidar, A. and Oberlander, T.F.
DOI: 10.1016/j.infbeh.2009.10.007Mind, Brain, and Education
Lombera, S. and Fine, A. and Grunau, R.E. and Illes, J.
DOI: 10.1111/j.1751-228X.2009.01079.xCanadian Journal of Psychiatry
Misri, S. and Kendrick, K. and Oberlander, T.F. and Norris, S. and Tomfohr, L. and Zhang, H. and Grunau, R.E.
DOI: 10.1177/070674371005500405Archives of Pediatrics and Adolescent Medicine
Oberlander, T.F. and Papsdorf, M. and Brain, U.M. and Misri, S. and Ross, C. and Grunau, R.E.
DOI: 10.1001/archpediatrics.2010.51IFMBE Proceedings
Doesburg, S.M. and Ribary, U. and Herdman, A.T. and Cheung, T. and Moiseev, A. and Weinberg, H. and Whitfield, M.F. and Synnes, A. and Liotti, M. and Weeks, D. and Grunau, R.E.
DOI: 10.1007/978-3-642-12197-5_57Early Human Development
Klein, V.C. and Gaspardo, C.M. and Martinez, F.E. and Grunau, R.E. and Linhares, M.B.M.
DOI: 10.1016/j.earlhumdev.2009.06.001Pain
Grunau, R.E. and Whitfield, M.F. and Petrie-Thomas, J. and Synnes, A.R. and Cepeda, I.L. and Keidar, A. and Rogers, M. and MacKay, M. and Hubber-Richard, P. and Johannesen, D.
DOI: 10.1016/j.pain.2009.02.014Clinical Journal of Pain
Holsti, L. and Grunau, R.E. and Oberlander, T.F. and Osiovich, H.
DOI: 10.1097/AJP.0b013e318158c5e5Archives of Disease in Childhood: Fetal and Neonatal Edition
Sherlock, R.L. and Synnes, A.R. and Grunau, R.E. and Holsti, L. and Hubber-Richard, P. and Johannesen, D. and Whitfield, M.F.
DOI: 10.1136/adc.2006.110726Early Human Development
Oberlander, T.F. and Grunau, R. and Mayes, L. and Riggs, W. and Rurak, D. and Papsdorf, M. and Misri, S. and Weinberg, J.
DOI: 10.1016/j.earlhumdev.2008.06.008Epigenetics
Oberlander, T.F. and Weinberg, J. and Papsdorf, M. and Grunau, R. and Misri, S. and Devlin, A.M.
DOI: 10.4161/epi.3.2.6034Infancy
Haley, D.W. and Grunau, R.E. and Oberlander, T.F. and Weinberg, J.
DOI: 10.1080/15250000802458682Pain
Holsti, L. and Grunau, R.E.
DOI: 10.1016/j.pain.2007.01.033Developmental Psychobiology
Mai, T.T. and Grunau, R.E. and Petrie-Thomas, J. and Haley, D.W. and Weinberg, J. and Whitfield, M.F.
DOI: 10.1002/dev.20204Journal of Pediatrics
Grunau, R.E. and Haley, D.W. and Whitfield, M.F. and Weinberg, J. and Yu, W. and Thiessen, P.
DOI: 10.1016/j.jpeds.2006.10.053Early Human Development
Holsti, L. and Weinberg, J. and Whitfield, M.F. and Grunau, R.E.
DOI: 10.1016/j.earlhumdev.2006.08.005Canadian Journal of Occupational Therapy
Holsti, L. and Grunau, R.E.
DOI: 10.1177/000841740707400306Archives of Pediatrics and Adolescent Medicine
Oberlander, T.F. and Reebye, P. and Misri, S. and Papsdorf, M. and Kim, J. and Grunau, R.E.
DOI: 10.1001/archpedi.161.1.22International Congress Series
Cepeda, I.L. and Grunau, R.E. and Weinberg, H. and Herdman, A.T. and Cheung, T. and Liotti, M. and Amir, A. and Synnes, A. and Whitfield, M.
DOI: 10.1016/j.ics.2006.12.090Developmental Medicine and Child Neurology
Grunau, R.E. and Whitfield, M.F. and Fay, T. and Holsti, L. and Oberlander, T. and Rogers, M.L.
DOI: 10.1017/S0012162206001010Psychoneuroendocrinology
Haley, D.W. and Weinberg, J. and Grunau, R.E.
DOI: 10.1016/j.psyneuen.2005.06.007Seminars in Fetal and Neonatal Medicine
Grunau, R.E. and Holsti, L. and Peters, J.W.B.
DOI: 10.1016/j.siny.2006.02.007Pediatrics
Alimenti, A. and Forbes, J.C. and Oberlander, T.F. and Money, D.M. and Grunau, R.E. and Papsdorf, M.P. and Maan, E. and Cole, L.J. and Burdge, D.R.
DOI: 10.1542/peds.2006-0525British Journal of Clinical Pharmacology
Kim, J. and Riggs, K.W. and Misri, S. and Kent, N. and Oberlander, T.F. and Grunau, R.E. and Fitzgerald, C. and Rurak, D.W.
DOI: 10.1111/j.1365-2125.2005.02538.xClinical Journal of Pain
Holsti, L. and Grunau, R.E. and Whifield, M.F. and Oberlander, T.F. and Lindh, V.
DOI: 10.1097/01.ajp.0000210921.10912.47Paediatrics and Child Health
Whitfield, M.F. and Grunau, R.E.
American Journal of Psychiatry
Misri, S. and Reebye, P. and Kendrick, K. and Carter, D. and Ryan, D. and Grunau, R.E. and Oberlander, T.F.
DOI: 10.1176/ajp.2006.163.6.1026Clinical Journal of Pain
Holsti, L. and Grunau, R.E. and Oberlander, T.F. and Whitfield, M.F. and Weinberg, J.
DOI: 10.1097/01.ajp.0000146163.30776.44Pediatrics
Rogers, M. and Fay, T.B. and Whitfield, M.F. and Tomlinson, J. and Grunau, R.E.
DOI: 10.1542/peds.2004-1603Pain
Grunau, R.E. and Holsti, L. and Haley, D.W. and Oberlander, T. and Weinberg, J. and Solimano, A. and Whitfield, M.F. and Fitzgerald, C. and Yu, W.
DOI: 10.1016/j.pain.2004.10.020Pediatrics
Oberlander, T.F. and Grunau, R.E. and Fitzgerald, C. and Papsdorf, M. and Rurak, D. and Riggs, W.
DOI: 10.1542/peds.2004-0420Early Human Development
Holsti, L. and Grunau, R.E. and Oberlander, T.F. and Whitfield, M.F.
DOI: 10.1016/j.earlhumdev.2004.08.002Clinical Journal of Pain
Grunau, R.E. and Linhares, M.B.M. and Holsti, L. and Oberlander, T.F. and Whitfield, M.F.
DOI: 10.1097/00002508-200403000-00003Pediatrics
Holsti, L. and Grunau, R.E. and Oberlander, T.F. and Whitfield, M.F.
DOI: 10.1542/peds.114.1.65Pediatrics
Grunau, R.E. and Weinberg, J. and Whitfield, M.F.
DOI: 10.1542/peds.114.1.e77Pediatrics
Grunau, R.E. and Whitfield, M.F. and Fay, T.B.
DOI: 10.1542/peds.2004-0932Early Human Development
Morison, S.J. and Holsti, L. and Grunau, R.E. and Whitfield, M.F. and Oberlander, T.F. and Chan, H.W.P. and Williams, L.
DOI: 10.1016/S0378-3782(03)00044-6Clinical Journal of Pain
Peters, J.W.B. and Koot, H.M. and Grunau, R.E. and De Boer, J. and Van Druenen, M.J. and Tibboel, D. and Duivenvoorden, H.J.
DOI: 10.1097/00002508-200311000-00003Archives of Pediatrics and Adolescent Medicine
Grunau, R.E. and Whitfield, M.F. and Davis, C.
DOI: 10.1001/archpedi.156.6.615Pediatric Research
Oberlander, T.F. and Grunau, R.E. and Fitzgerald, C. and Ellwood, A.-L. and Misri, S. and Rurak, D. and Riggs, K.W.
DOI: 10.1203/00006450-200204000-00008Clinics in Perinatology
Stevens, B. and Grunau, R.E.
DOI: 10.1016/S0095-5108(02)00023-4Pediatrics
Oberlander, T.F. and Grunau, R.E. and Fitzgerald, C. and Whitfield, M.F.
DOI: 10.1542/peds.110.3.570Clinics in Perinatology
Eckstein Grunau, R.
DOI: 10.1016/S0095-5108(02)00012-XInfant Mental Health Journal
Reebye, P.N. and Morison, S.J. and Panikkar, H. and Misri, S. and Grunau, R.E.
DOI: 10.1002/imhj.10025Journal of Developmental and Behavioral Pediatrics
Holsti, L. and Grunau, R.V.E. and Whitfield, M.F.
DOI: 10.1097/00004703-200202000-00002Primary Care - Clinics in Office Practice
Eckstein Grunau, R.
DOI: 10.1016/S0095-4543(02)00020-9Infant Behavior and Development
Eckstein Grunau, R. and Oberlander, T.F. and Whitfield, M.F. and Fitzgerald, C. and Morison, S.J. and Philip Saul, J.
DOI: 10.1016/S0163-6383(01)00065-0Archives of Pediatrics and Adolescent Medicine
Anand, K.J.S. and Aynsley-Green, A. and Bancalari, E. and Benini, F. and Champion, G.D. and Craig, K.D. and Dangel, T.S. and Fournier-Charri{\`e}re, E. and Franck, L.S. and Grunau, R.E. and Hertel, S.A. and Jacqz-Aigrain, E. and Jorch, G. and Kopelman, B.I. and Koren, G. and Larsson, B. and Marlow, N. and McIntosh, N. and Ohlsson, A. and Olsson, G. and Porter, F. and Richter, R. and Stevens, B. and Taddio, A.
DOI: 10.1001/archpedi.155.2.173Pediatrics
Grunau, R.E. and Oberlander, T.F. and Whitfield, M.F. and Fitzgerald, C. and Lee, S.K.
DOI: 10.1542/peds.107.1.105Clinical Journal of Pain
Morison, S.J. and Grunau, R.E. and Oberlander, T.F. and Whitfield, M.F.
DOI: 10.1097/00002508-200112000-00010Journal of Developmental and Behavioral Pediatrics
Grunau, R.E. and Whitfield, M.F. and Petrie, J.
DOI: 10.1097/00004703-200012000-00001Clinics in Perinatology
Whitfield, M.F. and Grunau, R.E.
DOI: 10.1016/S0095-5108(05)70026-9Clinical Journal of Pain
Grunau, R.E. and Holsti, L. and Whitfield, M.F. and Ling, E.
DOI: 10.1097/00002508-200003000-00007Pediatrics
Oberlander, T.F. and Grunau, R.E. and Whitfield, M.F. and Fitzgerald, C. and Pitfield, S. and Philip Saul, J.
DOI: 10.1542/peds.105.1.e6Pediatric Research
Oberlander, T.F. and Grunau, R.E. and Pitfield, S. and Whitfield, M.F. and Saul, J.P.
DOI: 10.1203/00006450-199904010-00011Journal of Developmental and Behavioral Pediatrics
Porter, F.L. and Grunau, R.E. and Anand, K.J.S.
DOI: 10.1097/00004703-199908000-00008Journal of Pediatrics
Scott, C.S. and Riggs, K.W. and Ling, E.W. and Fitzgerald, C.E. and Hill, M.L. and Grunau, R.V.E. and Solimano, A. and Craig, K.D.
DOI: 10.1016/s0022-3476(99)70163-0Journal of Child Psychology and Psychiatry and Allied Disciplines
Grunau, R.E. and Whitfield, M.F. and Petrie, J.
DOI: 10.1017/S0021963098002406Pain
Grunau, R.E. and Oberlander, T. and Holsti, L. and Whitfield, M.F.
DOI: 10.1016/S0304-3959(98)00046-3Research and Clinical Forums
Grunau, R.E.
Child and Adolescent Psychiatric Clinics of North America
Anand, K.S. and Grunau, R.E. and Oberlander, T.F.
DOI: 10.1016/s1056-4993(18)30274-8Pain
Hadjistavropoulos, H.D. and Craig, K.D. and Grunau, R.E. and Whitfield, M.F.
DOI: 10.1016/S0304-3959(97)00113-9Pain
Lilley, C.M. and Craig, K.D. and Grunau, R.E.
DOI: 10.1016/S0304-3959(97)00034-1Archives of Disease in Childhood: Fetal and Neonatal Edition
Whitfield, M.F. and Grunau, R.V.E. and Holsti, L.
DOI: 10.1136/fn.77.2.F85Journal of Obstetric, Gynecologic, & Neonatal Nursing
Stevens, B.J. and Johnston, C.C. and Grunau, R.V.E.
DOI: 10.1111/j.1552-6909.1995.tb02570.xPain
Grunau, R.V.E. and Whitfield, M.F. and Petrie, J.H. and Fryer, E.L.
DOI: 10.1016/0304-3959(94)90174-0Journal of Pediatric Psychology
Craig, K.D. and Hadjistavropoulos, H.D. and Grunau, R.V.E. and Whitfield, M.F.
DOI: 10.1093/jpepsy/19.3.305Pain
Grunau, R.V.E. and Whitfield, M.F. and Petrie, J.H.
DOI: 10.1016/0304-3959(94)90128-7Journal of Pediatric Psychology
Hadjistavropoulos, H.D. and Craig, K.D. and Grunau, R.V.E. and Johnston, C.C.
DOI: 10.1093/jpepsy/19.4.485Pain
Craig, K.D. and Whitfield, M.F. and Grunau, R.V.E. and Linton, J. and Hadjistavropoulos, H.D.
DOI: 10.1016/0304-3959(93)90162-IPain
Craig, K.D. and Whitfield, M.F. and Grunau, R.V.E. and Linton, J. and Hadjistavropoulos, H.D.
DOI: 10.1016/0304-3959(93)90106-YPain
Johnston, C.C. and Stevens, B. and Craig, K.D. and Grunau, R.V.E.
DOI: 10.1016/0304-3959(93)90132-9Nursing times
Grunau, R.
International Journal of Language & Communication Disorders
Grunau, R.V.E. and Kearney, S.M. and Whitfield, M.F.
DOI: 10.3109/13682829009011972Pain
Grunau, R.V.E. and Johnston, C.C. and Craig, K.D.
DOI: 10.1016/0304-3959(90)91142-6Pain
Grunau, R.V.E. and Craig, K.D.
DOI: 10.1016/0304-3959(87)90073-XJournal of clinical and experimental neuropsychology : official journal of the International Neuropsychological Society
Grunau, R.V. and Low, M.D.
DOI: 10.1080/01688638708410769Neuropsychologia
Grunau, R.V.E. and Purves, S.J. and McBurney, A.K. and Low, M.D.
DOI: 10.1016/0028-3932(81)90047-6Brain and Development
Dunn, H. and Crichton, J. and Grunau, R. and McBurney, A.K. and McCormick, A. and Robertson, A. and Schulzer, M.
DOI: 10.1016/S0387-7604(80)80008-8Journal of Educational Psychology
Grunau, R.V.
DOI: 10.1037/0022-0663.70.3.422The focus of my transdisciplinary research program is biobehavioural reactivity and infant neurodevelopment, broadly encompassing multiple aspects of infant arousal, self-regulation, attention, cognition and brain development in preterm and term born infants. My research program has been funded over the years primarily by the National Institutes of Health (NIH, USA) and the Canadian Institutes of Health Research (CIHR), as well as BCCHR and Michael Smith Foundation salary awards
Clinical practice of pain management in neonatal intensive care Unit (NICU) changed over the years from no concern with pain to widespread use of analgesia and sedation, however there are major gaps in knowledge of pain assessment in immature infants, and in long term effects of pain-related stress and pain medications. With the goal to improve pain assessment, we found that, along with recognized facial and heart rate changes, there are motor stress behaviors unique to premature infants (e.g. backward splaying of fingers). Conversely, other behaviors such as twitches and startles reflect sleep/waking state, and do not appear to be stress or pain cues. We established that greater exposure to neonatal pain-related stress in the NICU above and beyond multiple medical and neonatal risk factors, is associated with altered brain microstructure, white matter development, and cortical thinning on MRI and magnetoencephalography (MEG) oscillatory activity, as well as stress hormone (cortisol) expression, in turn related to cognitive development and internalizing behaviors, in infancy, early childhood and at school-age. Importantly, greater morphine exposure and sucrose use does not appear to prevent adverse long-term sequelae.
Notably, positive parent interaction style improved infant behavior, and ameliorated effects of early stress/pain exposure in very preterm infants.
In collaboration with geneticists in BCCHR, we identified some gene X environment (early pain/stress) interactions that modified long-term outcomes.
CIHR Operating Grant – Project: "Analgesia and sedation in the preterm neonate: brain development and outcome" (2014-2020)
Aubrey J. Tingle Prize, Michael Smith Foundation for Health Research 2021
Order of BC 2025
Jeffrey Lawson Award for Advocacy in Children’s Pain Relief, American Pain Society 2018
Rovee-Collier Mentor Award, International Society for Developmental Psychobiology, 2018
Outstanding Pain Mentorship Award, Canadian Pain Society 2019
Distinguished Career Award, International Association for the Study of Pain SIG Pain in Childhood 2019
Every day, our researchers work towards breakthroughs to transform the lives of kids in BC and around the world. Learn about our latest innovations and advancements in child health.

Each year in Canada, thousands of babies are born very prematurely — before 32 weeks gestation — and many require respiratory support in their first days and weeks of life. While this support is often life-saving, researchers are still learning how different types of respiratory care affect the developing brain.
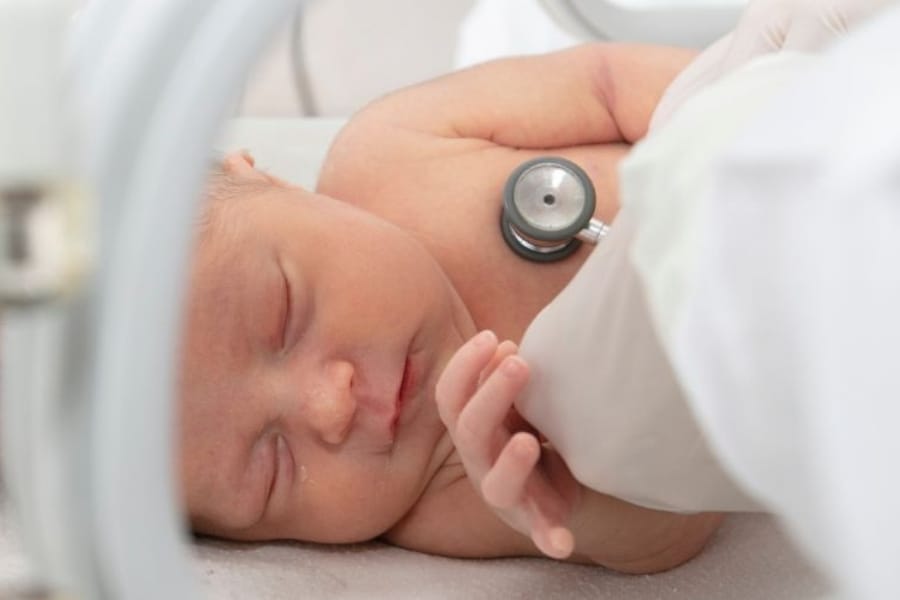
Very preterm babies — infants born two to four months early — require specialized care. Before they can safely go home, they need to spend time in the neonatal intensive care unit (NICU) until their organs are more developed. In the NICU, these babies are exposed to mildly painful procedures, including heel pricks for blood…

Very preterm babies have an increased risk of ongoing medical complications and neurodevelopmental issues throughout their lifespan. New research suggests that a specially designed epigenetic “clock” can indicate abnormally accelerated molecular aging, which may be associated with these long-term impacts and warrants further study.
At BC Children’s, we are making discoveries that save lives and transform health care for children in our province and around the world. Our research portfolio includes basic, clinical, population, and public health research.
EXPLORE OUR RESEARCH