Laura Sly
BSc, PhD, MSc
Investigator, BC Children's Hospital


International Journal of Molecular Sciences
Catherine M. Njeru and Bernard Ng and Sayeh Abdossamadi and Alima Suleimenova and Carmen Dolores De Luca and Vaishnavi Parthasarathy and Laura M. Sly and Gregor S. D. Reid and Chia Huan Ng and Kirk R. Schultz
DOI: 10.3390/ijms2705233803 / 2026
Immuno
Pang, Y.C.F. and Ma, W.J. and Menzies, S.C. and Sly, L.M.
DOI: 10.3390/immuno5030037Science
Ma, W.J. and Wang, C. and Kothandapani, J. and Luzentales-Simpson, M. and Menzies, S.C. and Bescucci, D.M. and Lange, M.E. and Fraser, A.S.C. and Gusse, J.F. and House, K.E. and Moote, P.E. and Xing, X. and Grondin, J.M. and Hui, B.W.-Q. and Clarke, S.T. and Shelton, T.G. and Haskey, N. and Gibson, D.L. and Martens, E.C. and Abbott, D.W. and Inglis, G.D. and Sly, L.M. and Brumer, H.
DOI: 10.1126/science.adk7633International Journal of Clinical Pharmacology and Therapeutics
Woodcock, B.G. and Braun, P. and Gasser, M. and Sly, L. and Lissner, R.
DOI: 10.5414/CP204615Biorxiv
Verdugo-Meza, A. and Gill, S.K. and Godovannyi, A. and Adur, M.K. and Barnett, J.A. and Estaki, M. and Ye, J. and Haskey, N. and Mehain, H. and Josephson, J.K. and Ishida, R. and Ghesquiere, C. and Sly, L.M. and Gibson, D.L.
DOI: 10.1101/2024.10.08.617317Trends in Immunology
Sly, L.M. and McKay, D.M.
DOI: 10.1016/j.it.2022.10.002International Journal of Clinical Pharmacology and Therapeutics
Oguz, S. and Buia, A. and Albers, B. and Suna, K. and Piroski, V. and Knapp, A. and Sly, L. and Dll, U. and Braun, P. and Woodcock, B.G.
DOI: 10.5414/CP204326Journal of the Canadian Association of Gastroenterology
Pang Y and Menzies S and Baksh S and Sly L
03 / 2021
Frontiers in cell and developmental biology
Luzentales-Simpson M and Pang YCF and Zhang A and Sousa JA and Sly LM
DOI: 10.3389/fcell.2021.612830 PubMed: 3361464502 / 2021
International journal of clinical pharmacology and therapeutics
Sly LM and Braun P and Woodcock BG
DOI: 10.5414/cp203890 PubMed: 3314101812 / 2020
Journal of the Canadian Association of Gastroenterology
Rees WD and Stahl M and Jacobson K and Bressler B and Sly LM and Vallance B and Steiner T
DOI: 10.1093/jcag/gwz047.20102 / 2020
Journal of the Canadian Association of Gastroenterology
Lisko D and Menzies S and Sly LM and Steiner T
DOI: 10.1093/jcag/gwz047.00902 / 2020
Journal of Crohn's and Colitis
Rees, W.D. and Stahl, M. and Jacobson, K. and Bressler, B. and Sly, L.M. and Vallance, B.A. and Steiner, T.S.
DOI: 10.1093/ECCO-JCC/JJZ194Journal of Leukocyte Biology
Rees, W.D. and Sly, L.M. and Steiner, T.S.
DOI: 10.1002/JLB.3MIR0120-567RThe Journal of experimental medicine
Hubbard, J.J. and Pyzik, M. and Rath, T. and Kozicky, L.K. and Sand, K.M.K. and Gandhi, A.K. and Grevys, A. and Foss, S. and Menzies, S.C. and Glickman, J.N. and Fiebiger, E. and Roopenian, D.C. and Sandlie, I. and Andersen, J.T. and Sly, L.M. and Baker, K. and Blumberg, R.S.
DOI: 10.1084/jem.20200359Pediatric Hematology and Oncology
Sly, L.M.
DOI: 10.1080/08880018.2020.1793850European Journal of Immunology
Kozicky, L.K. and Menzies, S.C. and Hotte, N. and Madsen, K.L. and Sly, L.M.
DOI: 10.1002/eji.201848014Journal of Leukocyte Biology
Monajemi, M. and Fisk, S. and Pang, Y.C.F. and Leung, J. and Menzies, S.C. and Ben-Othman, R. and Cai, B. and Kollmann, T.R. and Rozmus, J. and Sly, L.M.
DOI: 10.1002/JLB.5VMA0219-054RGut Microbes
Dobranowski, P.A. and Tang, C. and Sauv, J.P. and Menzies, S.C. and Sly, L.M.
DOI: 10.1080/19490976.2018.1560767Frontiers in Oncology
Liu, C.-C. and Leclair, P. and Pedari, F. and Vieira, H. and Monajemi, M. and Sly, L.M. and Reid, G.S. and Lim, C.J.
DOI: 10.3389/fonc.2019.00411Mucosal Immunology
Lo, Y. and Sauve, J.P. and Menzies, S.C. and Steiner, T.S. and Sly, L.M.
DOI: 10.1038/s41385-019-0191-zJournal of the Canadian Association of Gastroenterology
Monajemi M and Pang Y and Menzies S and Sly LM
DOI: 10.1093/jcag/gwy009.08002 / 2018
Journal of the Canadian Association of Gastroenterology
Lo Y and Sauv J and Menzies S and Sly LM
DOI: 10.1093/jcag/gwy008.00802 / 2018
Journal of the Canadian Association of Gastroenterology
Dobranowski P and Menzies S and Sly LM
DOI: 10.1093/jcag/gwy009.09402 / 2018
Cell Death and Disease
Leclair, P. and Liu, C.-C. and Monajemi, M. and Reid, G.S. and Sly, L.M. and Lim, C.J.
DOI: 10.1038/s41419-018-0601-2Journal of Leukocyte Biology
Dobranowski, P. and Sly, L.M.
DOI: 10.1002/JLB.3MIR0817-340RImmunology
Celiberto, L.S. and Graef, F.A. and Healey, G.R. and Bosman, E.S. and Jacobson, K. and Sly, L.M. and Vallance, B.A.
DOI: 10.1111/imm.12939Journal of Leukocyte Biology
Monajemi, M. and Pang, Y.C.F. and Bjornson, S. and Menzies, S.C. and van Rooijen, N. and Sly, L.M.
DOI: 10.1002/JLB.3VMA0118-019RFrontiers in Immunology
Kozicky, L.K. and Menzies, S.C. and Zhao, Z.Y. and Vira, T. and Harnden, K. and Safari, K. and Del Bel, K.L. and Turvey, S.E. and Sly, L.M.
DOI: 10.3389/fimmu.2018.02676European Journal of Immunology
Fidanza, M. and Seif, A.E. and Jo, S. and Kariminia, A. and Rolf, N. and Sly, L.M. and Grupp, S.A. and Reid, G.S.D.
DOI: 10.1002/eji.201646806Journal of Visualized Experiments
Kozicky, L. and Sly, L.M.
DOI: 10.3791/56689Biomaterials
Yang, H. and Kozicky, L. and Saferali, A. and Fung, S.-Y. and Afacan, N. and Cai, B. and Falsafi, R. and Gill, E. and Liu, M. and Kollmann, T.R. and Hancock, R.E.W. and Sly, L.M. and Turvey, S.E.
DOI: 10.1016/j.biomaterials.2016.09.032Cell Death and Disease
Liu, C.-C. and Leclair, P. and Monajemi, M. and Sly, L.M. and Reid, G.S. and Lim, C.J.
DOI: 10.1038/cddis.2016.176Gastroenterology
Ngoh, E.N. and Weisser, S.B. and Lo, Y. and Kozicky, L.K. and Jen, R. and Brugger, H.K. and Menzies, S.C. and McLarren, K.W. and Nackiewicz, D. and Van Rooijen, N. and Jacobson, K. and Ehses, J.A. and Turvey, S.E. and Sly, L.M.
DOI: 10.1053/j.gastro.2015.09.049Genes and Immunity
Ngoh, E.N. and Brugger, H.K. and Monajemi, M. and Menzies, S.C. and Hirschfeld, A.F. and Del Bel, K.L. and Jacobson, K. and Lavoie, P.M. and Turvey, S.E. and Sly, L.M.
DOI: 10.1038/gene.2015.30Seminars in Immunology
Kozicky, L.K. and Sly, L.M.
DOI: 10.1016/j.smim.2015.07.001European Journal of Immunology
Sharma, A.A. and Jen, R. and Kan, B. and Sharma, A. and Marchant, E. and Tang, A. and Gadawski, I. and Senger, C. and Skoll, A. and Turvey, S.E. and Sly, L.M. and Ct, H.C.F. and Lavoie, P.M.
DOI: 10.1002/eji.201444707Journal of Leukocyte Biology
Kozicky, L.K. and Zhao, Z.Y. and Menzies, S.C. and Fidanza, M. and Reid, G.S.D. and Wilhelmsen, K. and Hellman, J. and Hotte, N. and Madsen, K.L. and Sly, L.M.
DOI: 10.1189/jlb.3VMA0315-078RJournal of Allergy and Clinical Immunology
McKinnon, M.L. and Rozmus, J. and Fung, S.-Y. and Hirschfeld, A.F. and Del Bel, K.L. and Thomas, L. and Marr, N. and Martin, S.D. and Marwaha, A.K. and Priatel, J.J. and Tan, R. and Senger, C. and Tsang, A. and Prendiville, J. and Junker, A.K. and Seear, M. and Schultz, K.R. and Sly, L.M. and Holt, R.A. and Patel, M.S. and Friedman, J.M. and Turvey, S.E.
DOI: 10.1016/j.jaci.2013.10.045European Journal of Immunology
Weisser, S.B. and Kozicky, L.K. and Brugger, H.K. and Ngoh, E.N. and Cheung, B. and Jen, R. and Menzies, S.C. and Samarakoon, A. and Murray, P.J. and Lim, C.J. and Johnson, P. and Boucher, J.-L. and van Rooijen, N. and Sly, L.M.
DOI: 10.1002/eji.201343981Methods in Molecular Biology
Weisser, S.B. and McLarren, K.W. and Kuroda, E. and Sly, L.M.
DOI: 10.1007/978-1-62703-128-8-14Blood
Chaturvedi, A. and Araujo Cruz, M.M. and Jyotsana, N. and Sharma, A. and Yun, H. and Grlich, K. and Wichmann, M. and Schwarzer, A. and Preller, M. and Thol, F. and Meyer, J. and Haemmerle, R. and Struys, E.A. and Jansen, E.E. and Modlich, U. and Li, Z. and Sly, L.M. and Geffers, R. and Lindner, R. and Manstein, D.J. and Lehmann, U. and Krauter, J. and Ganser, A. and Heuser, M.
DOI: 10.1182/blood-2013-03-491571Weisser SB and McLarren KW and Kuroda E and Sly LM
DOI: 10.1007/978-1-62703-128-8_14 PubMed: 23179835Journal of Visualized Experiments
Weisser, S.B. and van Rooijen, N. and Sly, L.M.
DOI: 10.3791/4105European Journal of Immunology
Weisser, S.B. and Mclarren, K.W. and Voglmaier, N. and van Netten-Thomas, C.J. and Antov, A. and Flavell, R.A. and Sly, L.M.
DOI: 10.1002/eji.201041105Cancer Research
Ho, V.W. and Leung, K. and Hsu, A. and Luk, B. and Lai, J. and Shen, S.Y. and Minchinton, A.I. and Waterhouse, D. and Bally, M.B. and Lin, W. and Nelson, B.H. and Sly, L.M. and Krystal, G.
DOI: 10.1158/0008-5472.CAN-10-3973Journal of Leukocyte Biology
Weisser, S.B. and Brugger, H.K. and Voglmaier, N.S. and McLarren, K.W. and van Rooijen, N. and Sly, L.M.
DOI: 10.1189/jlb.0311124Journal of Immunology
Kuroda, E. and Antignano, F. and Ho, V.W. and Hughes, M.R. and Ruschmann, J. and Lam, V. and Kawakami, T. and Kerr, W.G. and McNagny, K.M. and Sly, L.M. and Krystal, G.
DOI: 10.4049/jimmunol.1002778American Journal of Pathology
McLarren, K.W. and Cole, A.E. and Weisser, S.B. and Voglmaier, N.S. and Conlin, V.S. and Jacobson, K. and Popescu, O. and Boucher, J.-L. and Sly, L.M.
DOI: 10.1016/j.ajpath.2011.03.018Experimental Hematology
Ruschmann, J. and Ho, V. and Antignano, F. and Kuroda, E. and Lam, V. and Ibaraki, M. and Snyder, K. and Kim, C. and Flavell, R.A. and Kawakami, T. and Sly, L. and Turhan, A.G. and Krystal, G.
DOI: 10.1016/j.exphem.2010.03.010Mediators of Inflammation
Ivison, S.M. and Khan, M.A.S. and Graham, N.R. and Shobab, L.A. and Yao, Y. and Kifayet, A. and Sly, L.M. and Steiner, T.S.
DOI: 10.1155/2010/652098Methods in Molecular Biology
Ho, V.W.H. and Sly, L.M.
DOI: 10.1007/978-1-59745-396-7_12Blood
Sly, L.M. and Hamilton, M.J. and Kuroda, E. and Ho, V.W. and Antignano, F.L. and Omeis, S.L. and Van Netten-Thomas, C.J. and Wong, D. and Brugger, H.K. and Williams, O. and Feldman, M.E. and Houseman, B.T. and Fiedler, D. and Shokat, K.M. and Krystal, G.
DOI: 10.1182/blood-2008-06-166082Blood
Heuser, M. and Sly, L.M. and Argiropoulos, B. and Kuchenbauer, F. and Lai, C. and Weng, A. and Leung, M. and Lin, G. and Brookes, C. and Fung, S. and Valk, P.J. and Delwel, R. and Lwenberg, B. and Krystal, G. and Humphries, R.K.
DOI: 10.1182/blood-2009-06-227603Handbook of Cell Signaling, Second Edition
Antignano, F. and Ruschmann, J. and Hamilton, M. and Ho, V. and Lam, V. and Kuroda, E. and Sly, L.M. and Krystal, G.
DOI: 10.1016/B978-0-12-374145-5.00134-0Journal of Immunology
Kuroda, E. and Ho, V. and Ruschmann, J. and Antignano, F. and Hamilton, M. and Rauh, M.J. and Antov, A. and Flavell, R.A. and Sly, L.M. and Krystal, G.
DOI: 10.4049/jimmunol.0900864Journal of Immunology
Locke, N.R. and Patterson, S.J. and Hamilton, M.J. and Sly, L.M. and Krystal, G. and Levings, M.K.
DOI: 10.4049/jimmunol.0803749Experimental Hematology
Argiropoulos, B. and Palmqvist, L. and Yung, E. and Kuchenbauer, F. and Heuser, M. and Sly, L.M. and Wan, A. and Krystal, G. and Humphries, R.K.
DOI: 10.1016/j.exphem.2008.02.011Infection and Immunity
Bishop, J.L. and Sly, L.M. and Krystal, G. and Finlay, B.B.
DOI: 10.1128/IAI.01596-07Journal of Immunology
Sly, L.M. and Kalesnikoff, J. and Lam, V. and Wong, D. and Song, C. and Omeis, S. and Chan, K. and Lee, C.W.K. and Siraganian, R.P. and Rivera, J. and Krystal, G.
DOI: 10.4049/jimmunol.181.6.3850Cancer Research
Krystal, G. and Sly, L. and Antignano, F. and Ho, V. and Ruschmann, J. and Hamilton, M.
DOI: 10.1158/0008-5472.CAN-07-0211Journal of Leukocyte Biology
Lee, J.S. and Nauseef, W.M. and Moeenrezakhanlou, A. and Sly, L.M. and Noubir, S. and Leidal, K.G. and Schlomann, J.M. and Krystal, G. and Reiner, N.E.
DOI: 10.1189/jlb.0906564Frontiers in Bioscience
Sly, L.M. and Ho, V. and Antignano, F. and Ruschmann, J. and Hamilton, M. and Lam, V. and Rauh, M.J. and Krystal, G.
DOI: 10.2741/2276Blood
Palmqvist, L. and Argiropoulos, B. and Pineault, N. and Abramovich, C. and Sly, L.M. and Krystal, G. and Wan, A. and Humphries, R.K.
DOI: 10.1182/blood-2005-12-007005Immunity
Rauh, M.J. and Ho, V. and Pereira, C. and Sham, A. and Sly, L.M. and Lam, V. and Huxham, L. and Minchinton, A.I. and Mui, A. and Krystal, G.
DOI: 10.1016/j.immuni.2005.09.003Rauh MJ and Sly LM and Kalesnikoff J and Hughes MR and Cao LP and Lam V and Krystal G
PubMed: 1549401511 / 2004
Biochemical Society Transactions
Rauh, M.J. and Sly, L.M. and Kalesnikoff, I. and Hughes, M.R. and Cao, L.-P. and Lam, V. and Krystal, G.
DOI: 10.1042/BST0320785Immunity
Sly, L.M. and Rauh, M.J. and Kalesnikoff, J. and Song, C.H. and Krystal, G.
DOI: 10.1016/j.immuni.2004.07.010Rauh MJ and Kalesnikoff J and Hughes M and Sly L and Lam V and Krystal G
PubMed: 1254670302 / 2003
Journal of Immunology
Lpez, M. and Sly, L.M. and Luu, Y. and Young, D. and Cooper, H. and Reiner, N.E.
DOI: 10.4049/jimmunol.170.5.2409Reviews of physiology, biochemistry and pharmacology
Kalesnikoff, J. and Sly, L.M. and Hughes, M.R. and Bchse, T. and Rauh, M.J. and Cao, L.P. and Lam, V. and Mui, A. and Huber, M. and Krystal, G.
Journal of Immunology
Sly, L.M. and Hingley-Wilson, S.M. and Reiner, N.E. and McMaster, W.R.
DOI: 10.4049/jimmunol.170.1.430Biochemical Society Transactions
Rauh, M.J. and Kalesnikoff, J. and Hughes, M. and Sly, L. and Lam, V. and Krystal, G.
DOI: 10.1042/bst0310286Experimental Hematology
Sly, L.M. and Rauh, M.J. and Kalesnikoff, J. and Bchse, T. and Krystal, G.
DOI: 10.1016/j.exphem.2003.09.011Infection and Immunity
Sly, L.M. and Guiney, D.G. and Reiner, N.E.
DOI: 10.1128/IAI.70.9.5312-5315.2002Journal of Biological Chemistry
Sly, L.M. and Lopez, M. and Nauseef, W.M. and Reiner, N.E.
DOI: 10.1074/jbc.M102876200Journal of Experimental Medicine
Hmama, Z. and Nandan, D. and Sly, L. and Knutson, K.L. and Herrera-Velit, P. and Reiner, N.E.
DOI: 10.1084/jem.190.11.1583Sly LM and Worobec EA and Perkins RE and Phibbs PV
PubMed: 830621011 / 1993
Canadian Journal of Microbiology
Sly, L.M. and Worobec, E.A. and Perkins, R.E. and Phibbs Jr., P.V.
DOI: 10.1139/m93-163Macrophages are critical effector cells in the inflammatory response. Classically activated macrophages initiate the innate immune response and direct the activity of the acquired immune response. Upon resolution of inflammation, macrophages convert to an anti-inflammatory phenotype called alternatively activated. Alternatively activated macrophages promote debris scavenging, tissue remodelling and wound healing. Intriguingly, macrophages can be manipulated to move back and forth between these two phenotypes. During inflammatory disorders, like inflammatory bowel disease, switching macrophages to an alternatively activated phenotype, could dampen down inflammation and reduce disease. We are currently assessing the macrophage phenotype present during inflammatory bowel disease and mechanisms that we could use to switch macrophages to an anti-inflammatory phenotype during disease.
L-arginine metabolism provides a key switch in macrophage phenotype. L-arginine can be metabolized by two pathways in macrophages. In classically activated macrophages during inflammation, the enzyme inducible nitric oxide synthase (iNOS) uses arginine to generate pro-inflammatory nitric oxide (NO). However, alternatively activated macrophages also express the enzyme arginase. Arginase metabolizes arginine leading to the production of polyamines, which promote cell growth, and proline, an essential component of collagen that can contribute to fibrosis. We are currently investigating the role of arginine metabolism by each of these pathways in inflammation and in fibrosis.
The src homology 2 domain-containing inositol 5'-phosphatase (SHIP) is a negative regulator of the phosphatidylinositol 3-kinase (PI3K) pathway. In vivo-differentiated SHIP deficient macrophages display a profoundly anergic phenotype and express phenotypic markers of alternative activation independent of canonical skewing by the Th2 cytokine, IL-4. SHIP protein levels are dramatically reduced when macrophages are skewed to an alternatively activate phenotype by IL-4 suggesting that increased PI3K activity may be required for canonical alternative activation of macrophages as well. We are currently characterizing the role of the PI3K pathway during canonical alternative activation of macrophages.
The G. Jeanette Thorbecke New Investigator Award from the Society for Leukocyte Biology (2012)
Canadian Association of Gastroenterology and Canadian Institutes of Health Research New Investigator Award (2009-2013)
Michael Smith Foundation for Health Research Scholar Award (2012-2021)
Every day, our researchers work towards breakthroughs to transform the lives of kids in BC and around the world. Learn about our latest innovations and advancements in child health.
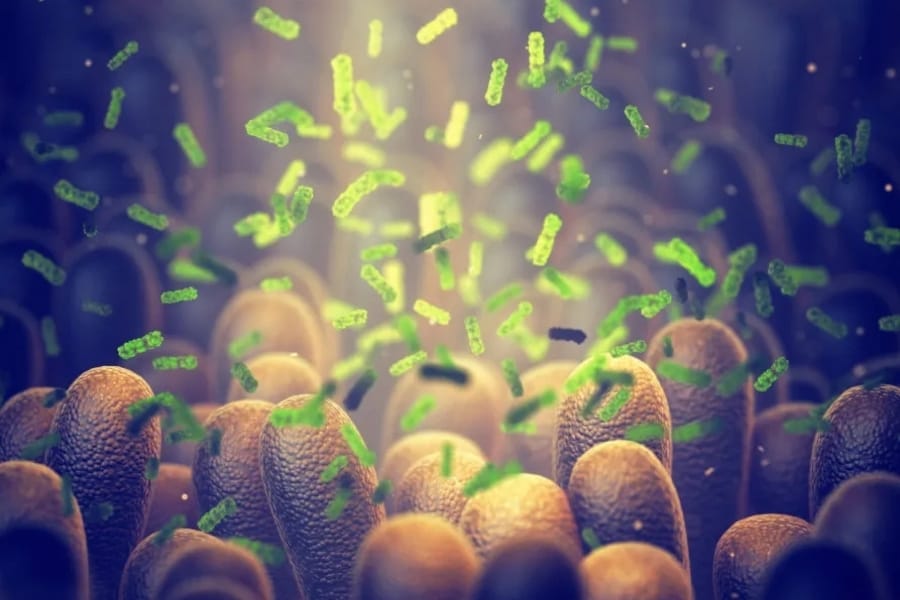
A new approach to drug design can deliver medicine directly to the gut in mice at significantly lower doses than current inflammatory bowel disease treatments.

Congratulations to the BC Children’s Hospital Research Institute (BCCHR), Women’s Health Research Institute (WHRI), and Vancouver Coastal Health Research Institute (VCHRI) investigators and their teams who were awarded funding through the Canadian Institutes of Health Research (CIHR) Spring 2024 Project Grant competition. Several of our research community members received around $5 million in new research…

Dr. Bruce Vallance, a researcher at BC Children’s Hospital, is a joint principal investigator on a new national research training program that will support the next generation of trainees and early-career researchers specializing in digestive health. The team, led by Dr. Nicola Jones from SickKids Hospital, as well as Dr. Vallance and Dr. Derek McKay…
At BC Children’s, we are making discoveries that save lives and transform health care for children in our province and around the world. Our research portfolio includes basic, clinical, population, and public health research.
EXPLORE OUR RESEARCH